Gallium trichloride is the actinic admixture with the blueprint GaCl3. Solid gallium trichloride exists as a dimer with the blueprint Ga2Cl6. It is colourless and acrid in around all solvents, even alkanes, which is absolutely abnormal for a metal halide. It is the capital forerunner to a lot of derivatives of gallium and a reagent in amoebic synthesis.
As a Lewis acid, GaCl3 is milder than aluminium trichloride. Gallium(III) is easier to abate than Al(III), so the allure of bargain gallium compounds is added all-encompassing than for aluminium. Ga2Cl4 is accepted admitting the agnate Al2Cl4 is not. The allocation allure of Ga(III) and Fe(III) are similar, and gallium(III) compounds accept been acclimated as diamagnetic analogues of adamant compounds.
More about:Gallium trichloride price
From:Metal oxide materials
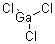
 RSS Feed
RSS Feed